ATTENDED AAN 2024
13th April-2024 To 18th-April-2024
#knowledgeshare #knowledgeupdate

World Parkinson’s Day 2024
Observed at KINS Institute of Neurosciences, Siliguri, India

































Clinically Isolated Syndrome
I feel honored and humbled in the same breath to be awarded with the Best Presentation for my co-authorship for the poster on "Clinically Isolated Syndrome Presenting with Writer's Cramp: A Rarity at Neuroimmunology-Movement Disorder Interface" by The PACTRIMS Scientific Committee 2023, Perth Australia.
I share this award with the other coauthor, Dr. Kadam Nagpal. Collaboration is the key…
Be#GreaterThan stroke
Stroke is the third leading cause of death and perhaps the leading cause of long term disabilities known to mankind. World-wide, 15 million people suffer stroke every year out of which 5 million die and another 5 million are significantly disabled.
Common risk factors are Hypertension, Diabetes, Dyslipidemia, Irregular heart rhythm, Heart failure, Smoking, Improper diet and Sedentary Lifestyle. Roughly 80 percent of all stroke are preventable by maintaining Blood pressure and Blood sugar in the Normal range, Good Diet and Exercise habits and saying no to smoke and alcohol. Regular health check up with special thrust to cardiac health, since the age as early as 25 years is a good practice. This world stroke day let’s pledge to work on preventive models more than other aspects of stroke, coz prevention is always better than cure.
Be#GreaterThan stroke
#prevention #stroke #disabilities #WSD
Dr. Swayam Prakash
MD, DM, SCE, FEBN, FICP , FRCP
Sr. Consultant & HOD - Neurology
Kins Institute of NeuroSciences
Multiple Sclerosis: A Smouldering Disease
Multiple sclerosis (MS) is a chronic neurological condition that affects approximately 2.8 million people worldwide. MS is a complex disease that affects the central nervous system, which includes the brain, spinal cord, and optic nerves. Although the cause of MS is still not fully understood, a combination of genetic and environmental factors are thought to play a role in its development.
MS Day is an international event that takes place on May 30th every year. The purpose of this day is to raise awareness about MS and to support individuals living with this condition.
Symptoms of MS:
MS can cause a wide range of symptoms that vary from person to person. The most common symptoms include:
1.Fatigue: Feeling tired or exhausted is a common symptom of MS. This can be due to the damage caused to the nervous system, which can make it harder for the body to function efficiently.
2.Cognitive Issues: MS can affect cognitive function, including memory, attention, and problem-solving. These symptoms can often be frustrating for individuals living with MS.
3.Vision Issues: MS can cause vision problems, including blurred vision, double vision, or loss of vision. These symptoms are caused by damage to the optic nerve.
4.Mobility Issues: MS can cause mobility issues, including difficulty walking, tremors, and weakness in the limbs. These symptoms can be due to damage to the neurons that control movement.
Causes of MS
The exact cause of MS is still not fully understood. However, researchers believe that a combination of genetic and environmental factors may play a role in its development.
Genetic Factors: MS is not directly inherited, but certain genes may increase a person's risk of developing the condition. Researchers have identified over 200 gene variants that are associated with MS.
Environmental Factors: Researchers have also identified several environmental factors that may increase a person's risk of developing MS. These factors include vitamin D deficiency, smoking, and exposure to certain viruses.
Treatment for MS
Although there is currently no cure for MS, there are several treatments that can help manage the symptoms of the condition. These treatments include:
1.Medications:There are several medications available that can help manage the symptoms of MS. These medications include corticosteroids, immunosuppressants, and disease-modifying therapies.
2.Physical Therapy: Physical therapy can help individuals with MS improve their mobility and reduce the risk of falls. Physical therapy can also help manage pain and improve energy levels.
3.Occupational Therapy: Occupational therapy can help individuals with MS learn new strategies to manage their daily activities. This can include learning how to conserve energy, adapt to physical limitations, and use assistive devices.
Summarisingly, MS is a chronic neurological condition that affects millions of people worldwide. Although the cause of MS is still not fully understood, there are several treatments available that can help manage the symptoms of the condition. MS Day is an important event that raises awareness about MS and provides support for individuals living with this condition. By increasing awareness of MS, we can help improve the quality of life for individuals affected by this condition.
#WorldMultipleSclerosisDay
#Multiplesclerosis
#Multiplesclerosisawareness
#KinsInstituteofNeurosciences
#KinsHealth
World Brain Day-22
Brain is perhaps the most complex thing known to mankind. It consists of roughly a hundred billion neurons with more than ten trillion connections helping it subserve functions as diverse as vegetative to sensori-motor to cognitive and behavioural. But Brain is at risk right since the time of conception in the mother’s womb. It’s exposed to the teratogenic effects of drugs, environmental toxins and radiations while in utero. Brain is the most vulnerable to the birth related injuries as well. During infancy and early childhood brain is highly susceptible to viruses, bacteria and other infectious agents leading to varying degree of injuries. Road traffic accidents are the leading cause of death and disabilities in young adults. Approximately 1.3 million people die each year due to road traffic accidents and between 20 and 50 million suffer non fatal injuries with permanent disabilities. Stroke and tumours are the leading causes of brain injuries in elderly. So Brain is at risk....
This world brain day, we have theme, “Brain health for all”. And there are five keys to achieve this, viz. Awareness, Prevention, Advocacy, Education and Access. Each one has its own importance but my thought goes with Prevention especially. I have some simple and pointed suggestions regarding prevention of brain insults at various stages of life.
A, planned pregnancy with timely prenatal checkups and compulsory institutionalised safe delivery practice can optimise foetal health and minimize birth related hypoxic brain injuries.
B, strict implementation of universal immunization program will effectively reduce deadly infective conditions of brain e.g., Meningitis, Encephalitis etc.
C, educating youths to live responsibly can reduce road traffic accidents, drug abuses and resulting brain damage.
And D, learning to respect aging while in youth by timely health check up and risk factors assessment can go a long way in preventing vascular brain insult like Stroke.
This world brain day, my Advocacy is for the equitable access of “Brain health for all”.
#BrainHealthForAll
#WorldBrainDay2022
#BrainHealth
#KinsInstituteofNeuroSciences
Dr. Swayam Prakash
MD, DM, SCE, FEBN, FRCP
Sr. Consultant & HOD - Neurology
Kins Institute of NeuroSciences
Multiple sclerosis (MS)
Multiple sclerosis (MS) is not an uncommon disease of the central nervous system (brain and spinal cord). Today, 2.8 million people around the world have MS.
MS is an inflammatory demyelinating condition. It is caused by damage to myelin. In MS, the loss of myelin affects the way nerves conduct electrical impulses to and from the brain. Common symptoms include blurred vision, weak limbs, tingling sensations, unsteadiness, loss of bladder bowel control, erectile dysfunction, memory problems, and fatigue.
Most people with MS are diagnosed between the ages of 20 and 40. MS is two to three times more common in women than in men. At present, there is no drug that can cure MS, but treatments are available which can modify the course of the disease favorably.
World MS Day is officially marked on 30 May. It brings the global MS community together to share stories, raise awareness, and campaign with everyone affected by multiple sclerosis (MS). We can choose to focus on a variety of angles when celebrating World MS Day, including:
* Challenging social barriers and stigma that can leave people affected by MS feeling lonely and isolated* Building communities that support and nurture people affected by MS* Promoting self-care and healthy living with MS*Lobbying decision-makers for better services and effective treatment for people with MS* Connecting people affected by MS to MS research.
‘I Connect, We Connect’ #MSConnection



Diary of a small town Neurologist
COVID-19 is perhaps the greatest disaster of all time, reported as a cluster of forty odd cases from Wuhan, China in December 2019 which culminated into a pandemic by March 2020, as declared by the WHO. And we were at war with an invisible enemy who was clever and powerful and was spreading at meteoric pace and only way to survive was to confine and conceal ourselves until we knew how to combat. With that came life to a screeching halt with its tangible repercussions on just everything around.
Healthcare arena also got changed dramatically with entire focus being shifted towards combating COVID-19, and perhaps rightly so but very rapidly it consumed most of the healthcare resources, leaving little for the Non-COVID patients. With the surge of second wave, the conditions got further worsened with literally no bed left for non-COVID emergencies like Acute Stroke, Status Epilepticus, Traumatic Brain Injuries, Severe Neuroimmunological conditions, Acute Myocardial Infarction etc. adding woes to the already suffering humanity. And smaller cities are rather badly hit. What pains me more is lack of preparedness and dearth of planning as how to overcome this situation.
In the current prevailing situation, as a Neurologist, I have following points to highlight with regards to Organization of care, Management of neurological disorders/symptoms, Management of neurological complications arising out of COVID-19 infections and Considerations for patients with chronic neurological conditions based on consensus statements of various scientific bodies:
Points to ponder regarding Organization of care
This is high time we realize that need for non COVID care is as much, if not more, as COVID care. This reminds me of a famous quote by Charles E. Hummel from Tyranny of the urgent- “Your greatest disaster is letting the urgent things crowd out the important.”
Hence unlike before, now we should focus on starting and further strengthening non-COVID hospitals/facilities/wings for non-COVID emergencies where standardized treatment for various neurological/neurosurgical emergencies could be started without delay while taking due care to prevent contamination or spread of infections.
Teleconsultation should be encouraged for routine patients not requiring admission; however, those patients who may require in person consultation should be picked up from there and advised accordingly.
Postpone all elective EEG/EMG/imaging investigations unless urgent and likely to impact clinical decision making and treatment plans and contamination prevention guidelines must be adhered to if at all it’s necessary.
Special management issues
Acute Stroke is a time sensitive neurological condition therefore properly selected patients of acute ischemic stroke should receive IV thrombolysis/ endovascular treatment in timely fashion.
Treatment of Convulsive Status Epilepticus shoud receive top urgent priority as per recent ILAE guidelines.
While using Cell depleting therapy (Rituximab, Ocrelizumzb, Alemtuzumab etc.) for neuroimmunological conditions, risk-benefit ratio must be properly weighed against each other as they increase susceptibility to contract infection for several months after infusion.
Regarding use of IVMPS/IVIG/PLEX, though they do not pose any additional risk yet should not be started if there are signs of active infection
Neurological complications arising out of COVID 19 infection
We should also be mindful that severe neurological complications can occur in COVID-19 patients during hospitalisation, such as Seizures, Encephalopathy, Encephalitis and Cerebrovascular events including Ischaemic Stroke or Intracerebral Haemorrhage.
Prolonged ICU admission may cause development of Multifactorial Encephalopathy, Critical Illness Neuropathy and Myopathy.
Post ICU-care syndrome develop in ICU-survivors in the form of cognitive impairment, psychiatric and/or physical disability.
There may be a higher risk of sub acute neurological complications, including GBS and other autoimmune diseases such as Necrotizing Encephalitis following COVID-19 infection.
Therefore, Neurologists must be kept in the loop while treating COVID-19 patients to detect and treat neurological symptoms and disorders at the earliest.
Care for patients with chronic neurological disorders is specially challenging
Patients on immunosuppressive medications should practice extra vigilant social distancing, including avoiding public gatherings/crowds and avoiding crowded public transport etc.
Patients of Parkinson’s disease may be particularly vulnerable to respiratory infections or pneumonia due to limited respiratory capacity related to reduced mobility of their thoracic cage. Therefore, it is important to counsel such patients to undertake all precautions for reducing exposure risk.
Epileptic patients may have drug–drug interactions between antiepileptic drugs (AEDs) and newer treatment options for COVID-19 including antiviral, immunomodulatory and immunosuppressive drugs. Hence, dose adjustments of AEDs or COVID-19 treatment might be necessary
Besides above specific measures, COVID-appropriate behaviours are the keys in keeping pandemic at bay.
There are many diseases but, in a sense, there is just one health. STAY SAFE!
Myasthenia Gravis
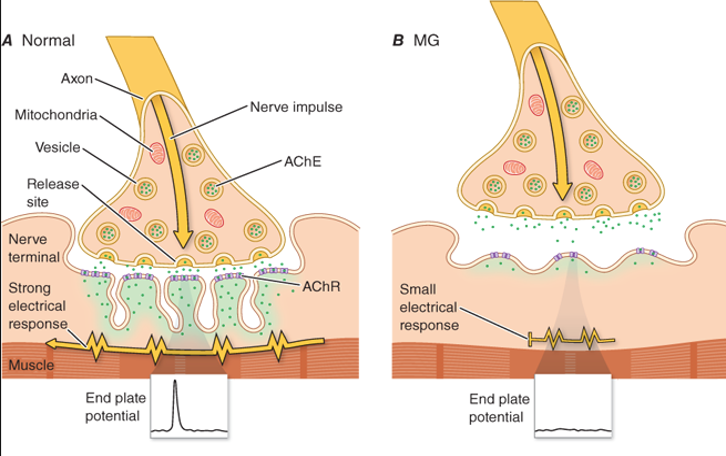
Myasthenia gravis is an acquired organ specific autoimmune disorder of neuromuscular transmission resulting from antibodies directed against the Acetylcholine receptor (AChR), Muscle specific tyrosine kinase (MuSK) and Lipoprotein related protein 4 (LRP4).
The incidence of myasthenia is slightly greater in women than in men (3:2) and the estimated prevalence is around 20 per 100000 population.
The clinical hallmark of myasthenia gravis is fluctuating weakness and abnormal fatigability of voluntary skeletal muscles that improve with rest. Drooping of eyelid, double vision, difficulty swallowing, slurring of speech or change of voice, and proximal limb weakness are the commonest symptoms. Occasionally, patients may present with difficulty breathing due to respiratory muscle weakness known as myasthenic crisis. Stressful life events, viral infections, pregnancy and delivery may precipitate the development of myasthenic crisis.
Diagnosis is mainly clinical supported by electrophysiologic (RNST,SFEMG) and lab tests (AChR ab). MG is associated with other autoimmune diseases in about 30% of cases, eg. thyroid diseases, RA, SLE etc.
Acetylcholine esterase inhibitors, immunosuppressive/immunomodulating drugs, plasma exchange (PLEX) and thymectomy are various treatment modalities in the selected set of patients with fairly good results.
Although the number of patients with MG continues to rise, it is still a rare disease. Further epidemiological research with a view to establish population registries and to estimate economic impact of disease on population as well as quality of life of patients with MG is needed.
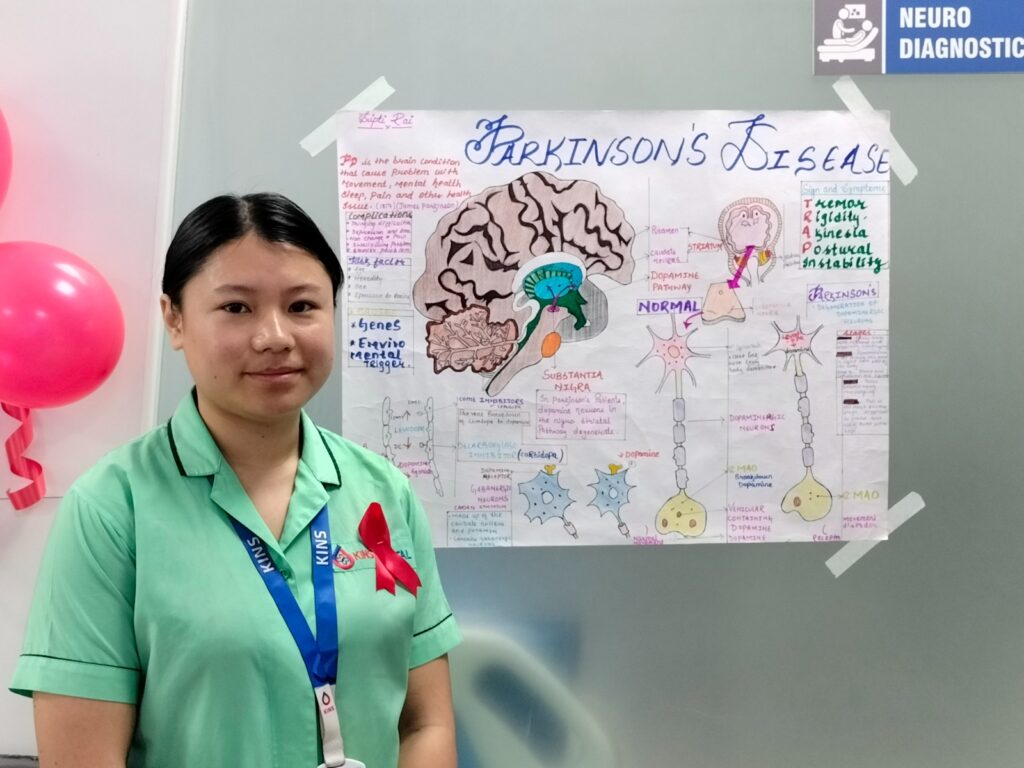
World Parkinson’s Day
[16:47, 12/04/2021] Anindya Da: World Parkinson’s Day is celebrated each year on april 11 in the honour of James Parkinson, a great neurologist, researcher and activist, who was born on this date in the year 1755. He described the cardinal features of Parkinson’s disease (PD) in his famous write up “an essay on shaking palsy” in the year 1817. Later on, Dr. Jean-Martin Charcot named this disease after James Parkinson, calling it “Maladie de Parkinson” (Parkinson’s disease) as a gesture of acknowledgement and respect. April is celebrated as parkinsons awareness month where in doctors, nurses, physiotherapists, speech therapists, behavioural therapists, occupational therapists, support groups, many government and non government organizations worldwide come together in creating positivity and hope in the life of patients with PD. This year, the theme is, “unite for Parkinsons”, a call to action that urges people to stand for patients suffering from Parkinson’s and their loved ones and to help them in all possible ways. The idea is to spread the word and engage people and organizations in making life of patients with Parkinson disease easy and society more responsive towards them.
There are nearly ten million people living with Parkinsons disease worldwide, men being 1.5 times more affected than women. The incidence and prevalence increases with advancing age, around 1% of people above 65 years of age being affected from PD. There is no homogeneous and large epidemiological data available from India, however, sectoral data suggest a crude prevalence rate of 14.1 per 100,000 population in nothern India (Kashmir), 27 per 100,000 in southern part of India (Bangalore), 16.1 per 100,000 in eastern India (rural Bengal), and 328.3 per 100,000 among a population of 14,010 Parsis living in colonies in Mumbai, western India.
Parkinson’s disease is a chronic progressive neurodegenerative disorder characterized by early prominent loss of dopaminergic neurons in the substantia nigra pars compacta (SNpc) and wide spread presence of alpha synuclein (aSyn), an intracellular protein. Dopamine deficiency in the basal ganglia leads to the classical Parkinsonian motor symptoms viz, bradykinesia, tremor, rigidity and later postural instability. PD is also associated with non-motor symptoms viz, sleep disorders, hyposmia, autonomic dysfunctions, cognitive impairment, mood disorders and pain, which may precede motor symptoms by more than a decade. These non-motor symptoms become troublesome symptoms in the later stages of PD. Currently, the mainstay of PD management is pharmacological therapy, which include levodopa/ carbidopa, dopamine agonists (both ergot and non-ergot types), monoamine oxidase-B (MAO-B) inhibitors, injectable dopamine agonist (apomorphine), catechol-O-methyltransferase (COMT) inhibitors, N-methyl-D-aspartate (NMDA) receptor inhibitors, and anti-cholinergics. Dopaminergic therapy is highly effective in bradykinesia and rigidity but monoamine MAO B inhibitors are only moderately effective. Dopamine agonists and levodopa help to reduce disease progression and disability to an extent. Tremor responds to anticholinergic drugs like trihexyphenidyl but has a poor and inconsistent response to dopamine replacement therapy. However, these symptomatic therapies have major limitations in advanced disease. Many disabling features develop later in the course of the disease including non-motor symptoms, dopamine resistant motor symptoms and motor complications of long-term dopamine therapy. In the later stages of PD, drug delivery can be supplemented via alternative routes (e.g., intrajejunal infusions, subcutaneous injections or transdermal patches). Continued motor fluctuations and dyskinesias may be indications to evaluate the patient’s candidacy for deep brain stimulation (DBS). Although there have been remarkable advances in the medical and surgical treatment for PD, definitive disease modifying therapy is still eluding. However, researchers are hopeful that they will be able to identify the potential targets for disease modification in near future.
Non-pharmacological therapies viz, exercise, education, support groups, speech therapy and nutrition, are equally important and play a great role, if started early on in the disease course, in retarding the progression of disease and improving the quality of life of PD patients.
In the prevailing Covid-19 situation, patients with parkinsons disease are at higher risk because, they are usually aged population with waning immunity. Respiratory muscle rigidity as well as poor respiratory excursion makes them further vulnerable to ventilatory failure in the event of Covid-19 infection. Covid-19, like any other infection, could also lead to the worsening of parkinsonian motor symptoms like bradykinesia and or dyskinesias.
Therefore all precautions advised to avoid Covid-19 infection need to be followed by all PD patients, including social/physical distancing and 20-seconds hand washing. In severely affected patients needing ventilatory support, non-oral dopamine replacement strategy should be resorted to eg., liquid levodopa (via nasogatric tube or PEG), transdermal rotigotine patches, or apomorphine injection/infusion. Non motor symptoms especially; anxiety, depression and fatigue must also be enquired about and properly addressed. Drug – drug interactions especially, patients on MAO-B inhibitors with cough- cold remedies should be borne in mind while prescribing. Other drugs being tested and tried against Covid-19 viz., lopinavir, ritonavir, remdesevir, tociizumab, hydroxychloroquine etc have not shown any significant interaction with PD drugs, so far. Further, Amntadine, which is an ativiral agent used for PD, needs to be evaluated for its potential anti-Covid-19 effect. Hence amidst Covid-19, care for PD patients warrants further attention.
“While there are many diseases, there is, in a sense, only one health”. Park’s Text Book of PSM
Rare Disease

Rare #Many #Strong #Proud
There is no precise definition of RARE DISEASE. WHO defines a disease to be rare if it affects 1 or less per 1000 population. However, European definition for rare disease is, when it affects less than 1 in 2000 people. Considering the population of India the suggested threshold for India for a disease to be defined as rare is 1 in 10,000.
There are over 300 million people living with one or more of over 7000 identified rare diseases around the world. Each rare disease may only affect a handful of people, scattered around the world, but they add up to a formidable number. Rare diseases currently affect 3.5% - 5.9% of the population worldwide. 72% of rare diseases are genetic whilst others are the result of infections (bacterial or viral), allergies and environmental causes, or are degenerative and proliferative and roughly 70% of those genetic rare diseases start in early childhood.
According to an estimate, about a third of rare diseases are due to some Neutological disorders, eg. Antibody mediated encephalitis, ALS, Ataxia telangiectasia, Ataxia with occulomotor apraxia, Autosomal dominant Leucodystrophies, BMD, Benign essential blepharospasm, Brown Sequard syndrome, CADASIL, Chanelopathies, CMTs, Congenital Myasthenic syndrome, Dermatomyositis, Distal Myopathies, Dopa resrponsive dystonia, Episodic ataxias, Friedreich ataxia, Familial hemiplegic migraine, Fronto-temporal dementia, GBS, Hereditary spastic paraparesis, Huntington’s Disease, Inborn errors of metabolism, Joubert syndrome, Lambert eaton syndrome, Leucodystrophies, LGMDs, Mitochondrial disorders, Myasthenia Graves, Marinesco sjogren Syndrome, Meige Syndrome, Meralgia Paresthetica, Miller-Fischer Syndrome, MSA, Myotonia congenita, Myotonic dystrophy, Narcolepsy, NBIA, Neuroblastoma, Neurofibromatosis, NMOSD, Paroxysmal dyskinesias, Post polio syndrome, SCAs, Spastic paraplegia, Status epilepticus, Wilson Disease etc. to name a few. Hence the role of Neurologists cannot be over emphasised in the precise diagnosis and appropriate management of these conditions.
Rare diseases are characterised by a broad range of disorders and symptoms that vary not only from disease to disease but also from patient to patient suffering from the same disease. Varied clinical presentation and lack of scientific knowledge often lead to misdiagnosis. Lack of treatment/cure further adds to the woes.
Rare Disease Day takes place on the last day of February each year, a month with rare number of days. Rare Disease Day raises awareness for the 300 million people living with rare diseases around the world and their families and carers. The campaign targets primarily the general public and also seeks to raise awareness amongst policy makers, public authorities, industry representatives, researchers, health professionals and anyone who has a genuine interest in rare diseases.
The long-term cause of the Rare Disease Day campaign is to achieve equitable access to diagnosis, treatment, health and social care and social opportunity for people affected by a rare disease.
“Let’s be a part of this global noble cause…..”
Dementia or Neurocognitive disorders
The term Dementia is derived from Latin stock, meaning without mind. According to DSM fourth edition, text revision, dementia refers to multiple cognitive deficits that involves memory impairment, and one or more of the following, viz. language disturbance (aphasia), impairment in carrying out skilled motor activities despite intact motor functions (apraxia), deficits in recognizing familiar persons or object despite intact sensory function (agnosia), impairment in planning, initiating, organizing, and abstract reasoning (executive dysfunction). DSM-5 replaced the term dementia with neurocognitive disorder, major or minor. It also obviated the need of memory impairment to qualify the definition of the disorder.
Alzheimer’s disease (AD) is the most common form of dementia and accounts for nearly 60-70% of all cases. As per a current estimate AD affects over 35 million people worldwide.
Age is perhaps the most important risk factor, as prevalence rate doubles every five years after age 65. Other important risk factors are postmenopausal women, lower intelligence, fewer year of education, traumatic brain injury, and many medical and psychiatric conditions eg. Diabetes, hypertension, stroke, MI, dyslipidemia, obesity, chronic stress, depression etc. There are some genetic risk factors also like Presenilin 1&2, APP, APOE genes etc.
The evolving history of slowly progressive cognitive decline involving memory and other domains is the most important diagnostic clue which is aided by neuropsychological battery (MMSE, MOCA, FAB, Lobar function), neuroimaging ( MRI, amyloid PET, FDG-PET) and biomarker (Abeta /tau in CSF) to arrive at a proper diagnosis.
AD risk may be decreased by regular exercise, mental stimulation, socialization and better dietary habits with regular intake of low calorie, low fat diet rich in omega 3 fatty acids/DHA, vitamins (folate, B12, B3, C,E) and antioxidants; and regular servings of fruits and vegetables.
Donepezil, Rivastigmine, Galantamine, Mementine and Caprylidine triglyceride are FDA approved medications for the treatment of AD. Cognitive rehabilitation, involving problem solving and attention skills, has also been suggested to slow the progression of the disease and facilitate gain in skills.
So timely recognition of symptoms by the caregiver and physicians lie to the core of a ready diagnosis and institution of treatment and rehabilitation which help patients gain the lost skills and lead a better and graceful life with fair degree of independence.
